What Is CDISC and Why Should Sponsors Care?
Table of Contents
CDISC (Clinical Data Interchange Standards Consortium) develops global data standards for clinical research. If you’re submitting data to the FDA or EMA, CDISC standards aren’t optional — they’re required.
The CDISC standards ecosystem
- CDASH (Clinical Data Acquisition Standards Harmonization) — defines standard CRF fields and structures for data collection
- SDTM (Study Data Tabulation Model) — the format for submitting raw clinical data to regulators
- ADaM (Analysis Data Model) — the format for analysis-ready datasets used in statistical programming
- SEND (Standard for Exchange of Nonclinical Data) — for preclinical/animal study data
- Define-XML — metadata standard that describes the structure and content of submission datasets
Why sponsors should care
The FDA requires SDTM and ADaM formats for all NDA, BLA, and IND submissions. The EMA and PMDA have similar requirements. Non-compliant submissions trigger technical rejection — meaning your data goes back to the end of the review queue.
But compliance isn’t the only reason to invest in CDISC:
- Faster submissions. Well-structured data speeds FDA review by making it easier for reviewers to run their standard analyses
- Cross-study analysis. CDISC-standardized data enables meta-analyses and integrated summaries across your clinical program
- Reduced programming costs. Starting with CDASH-aligned CRFs means less data mapping and programming to get to SDTM/ADaM at the end
Common mistakes
- Retrofitting standards at submission time. Trying to map non-standard data into SDTM after database lock is expensive and error-prone. Start with CDASH-aligned CRFs from day one.
- Ignoring controlled terminology. CDISC has published controlled terminology for hundreds of variables. Using non-standard terms creates mapping headaches.
- Choosing EDC systems that don’t support CDISC. Your EDC should generate CDISC-compliant datasets natively. See our EDC selection guide.
Getting started
If your organization is new to CDISC, start here:
- Map your CRFs to CDASH domains before your first study builds
- Build a company-level CDISC standards library that can be reused across studies
- Train your data management and programming teams on SDTM and ADaM
- Choose technology that enforces CDISC standards at the point of data capture
See how Clincove’s EDC supports CDISC standards from data capture through submission.
Continue Reading

Decentralized Trials in 2026: What’s Working and What Isn’t
DCTs promised to revolutionize clinical research. Three years post-pandemic, we look at what’s actually delivering resul
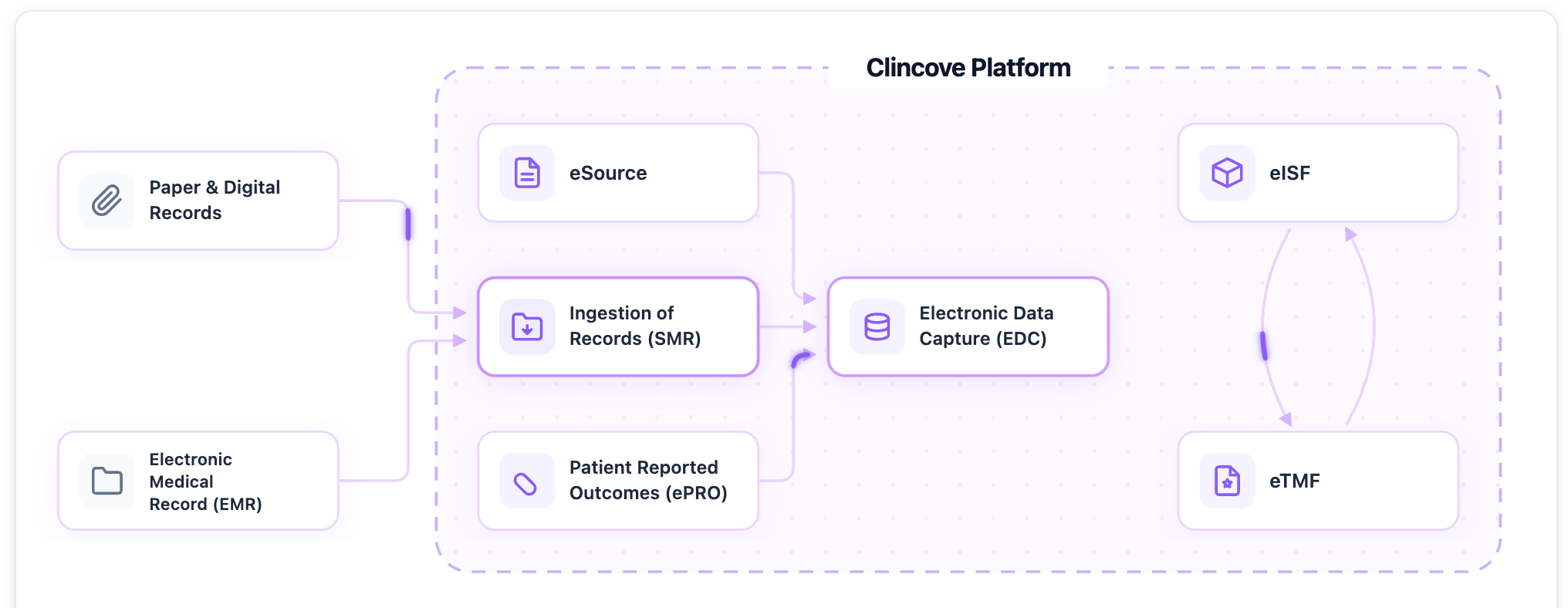
Protocol AI: How We Built CRF (Case Report Forms) Auto-Generation - Modernizing EDC
A behind-the-scenes look at how Clincove’s Protocol AI reads protocol documents and automatically generates case report

5 Red Flags in Your Clinical Trial Budget (And How to Fix Them)
Most trial budgets have hidden cost overruns baked in from day one. Learn to spot the five most common red flags before
Simplify clinical operations
Ready to modernize your clinical trials?
See how Clincove unifies EDC, eISF, eTMF, and eSource into one platform -- deploy in weeks, not months.