Clinical Trial Budget Planning: The Complete 2026 Guide

Table of Contents
Clinical trial budgets are notoriously difficult to plan and even harder to control. Whether you’re a small biotech planning your first Phase I or a large pharma sponsor managing a global Phase III, understanding the full cost landscape is critical for making informed decisions.
This guide covers every major budget category and practical strategies for 2026.
The big picture: what trials actually cost
Average per-study costs vary enormously by phase and therapeutic area, but here are benchmark ranges:
- Phase I: $2–5 million (healthy volunteer or first-in-human)
- Phase II: $10–30 million (proof of concept, dose-finding)
- Phase III: $30–100+ million (pivotal, registration-enabling)
- Phase IV: $5–20 million (post-marketing, REMS studies)
Within these totals, the cost per patient typically ranges from $15,000–$50,000 for Phase II and $25,000–$75,000 for Phase III, depending on complexity.
Major budget categories
Site costs (typically 40–60% of total)
- Per-patient grants. The largest single line item. Covers all site activities per enrolled subject — screening, visits, procedures, data entry, and follow-up.
- Start-up costs. Site activation fees, IRB submission fees, regulatory document preparation, and site training.
- Lab and procedure pass-throughs. Central lab fees, imaging, ECGs, and other procedure costs that are billed separately from per-patient grants.
CRO costs (if applicable, typically 20–30%)
- Monitoring fees. CRA time for site visits, remote monitoring, and data review.
- Data management. Clinical data management, programming, and query resolution.
- Project management. Oversight, coordination, and regulatory support.
- Medical monitoring. Physician review of safety data and medical queries.
Technology costs (typically 5–15%)
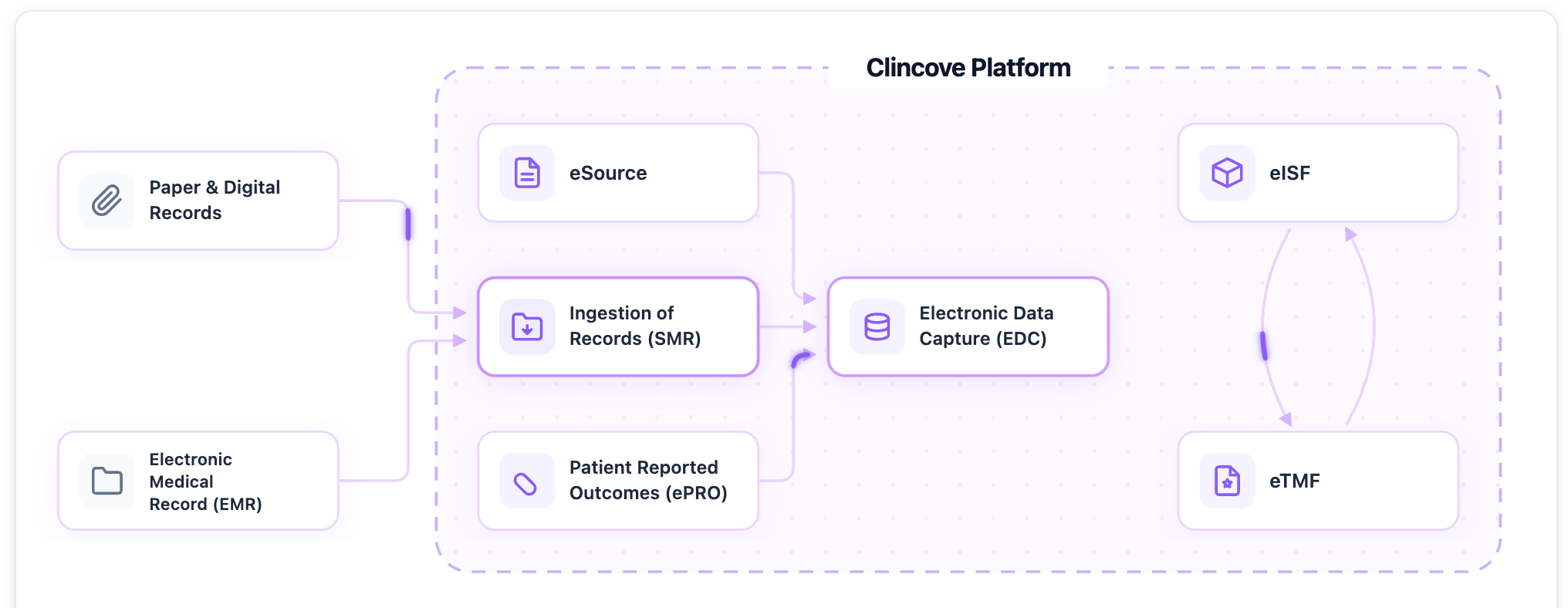
- EDC and data capture. License fees, setup, CRF design, and hosting.
- Document management. eTMF, eISF, and regulatory compliance systems.
- RTSM. Randomization and trial supply management system setup and maintenance.
- Safety database. Pharmacovigilance system for AE/SAE management.
Hidden costs to watch for
- Protocol amendments. Each Amendment costs an estimated $500K+ in direct costs and 3+ months of delay. Read about budget red flags.
- Screen failures. Sites spend significant time screening patients who ultimately don’t qualify. High screen failure rates (>50%) dramatically increase per-enrolled-patient costs.
- Data reconciliation. Data silos create manual reconciliation work that’s expensive and time-consuming.
- Change orders. CRO scope changes mid-trial almost always cost more than the original estimate.
- Extended timelines. Every month a trial runs over schedule costs in staff time, site fees, and opportunity cost.
Strategies to reduce trial costs in 2026
- Use a unified platform. Replacing 5+ point solutions with one integrated platform reduces technology licensing costs, eliminates integration expenses, and cuts training time.
- Adopt eSource. eSource reduces queries by 60%+, which directly reduces CRO data management costs and accelerates database lock.
- Implement risk-based monitoring. RBM focuses monitoring resources where they matter most, reducing CRA travel costs by 30–50%.
- Invest in protocol design. Spending more time (and using AI tools) on protocol design upfront reduces amendments, screen failures, and operational complexity.
- Negotiate milestone-based CRO contracts. Align CRO incentives with trial outcomes, not just activity volume.
Planning your next trial budget? See how Clincove reduces total trial costs through platform consolidation and operational efficiency.
Continue Reading

Decentralized Trials in 2026: What’s Working and What Isn’t
DCTs promised to revolutionize clinical research. Three years post-pandemic, we look at what’s actually delivering resul
Protocol AI: How We Built CRF (Case Report Forms) Auto-Generation - Modernizing EDC
A behind-the-scenes look at how Clincove’s Protocol AI reads protocol documents and automatically generates case report

5 Red Flags in Your Clinical Trial Budget (And How to Fix Them)
Most trial budgets have hidden cost overruns baked in from day one. Learn to spot the five most common red flags before
Simplify clinical operations
Ready to modernize your clinical trials?
See how Clincove unifies EDC, eISF, eTMF, and eSource into one platform -- deploy in weeks, not months.