eTMF vs. eISF: What’s the Difference, Why It Matters & How to Automate Transfer & Filing
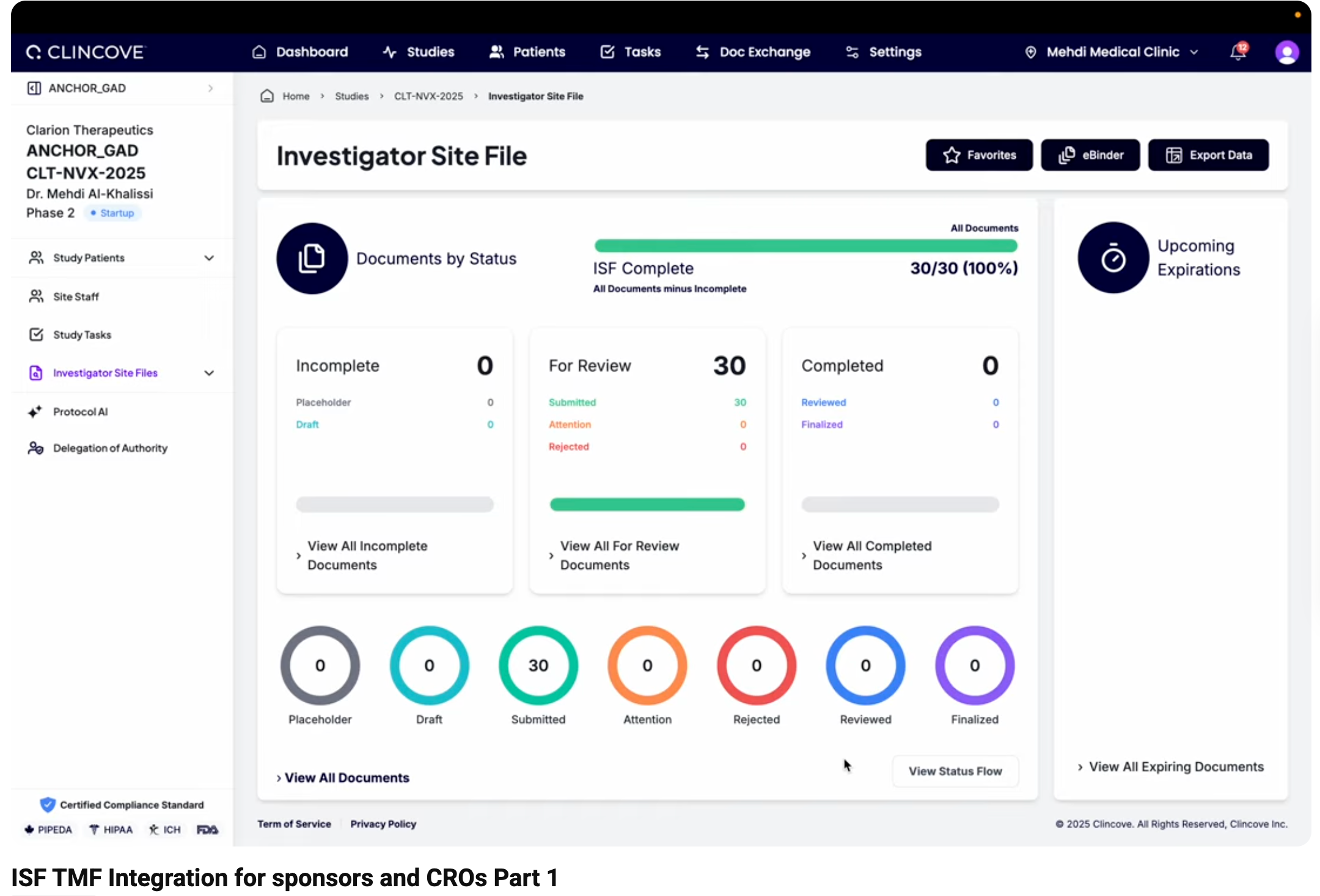
Table of Contents
The eTMF and eISF are both essential document management systems in clinical trials, but they serve different audiences and different purposes. Understanding the distinction helps sponsors, CROs, and sites choose the right approach.
What’s an eTMF?
The electronic Trial Master File (eTMF) is the sponsor-side document repository. It contains all essential documents for a clinical trial as defined by ICH GCP and the DIA TMF Reference Model.
The eTMF is owned and maintained by the sponsor (or delegated to a CRO). It must be audit-ready and available for regulatory inspection throughout the trial and for the required retention period.
What’s an eISF?
The electronic Investigator Site File is the site-side document repository. It contains the same essential document types, but from the site’s perspective — signed delegation logs, local IRB approvals, site-specific training records, and the investigator’s copies of the protocol and amendments.
The eISF is maintained by the site and must be available for monitor review and regulatory inspection.
Key differences
- Ownership: eTMF belongs to the sponsor/CRO; eISF belongs to the site
- Scope: eTMF covers the entire trial across all sites; eISF covers one site’s documents
- Access: eTMF is accessed by sponsor, CRO, and regulatory staff; eISF is accessed by site staff and monitors
- Regulatory basis: Both are required by ICH GCP, but the DIA TMF Reference Model provides more detailed structure for the eTMF
Why integration matters
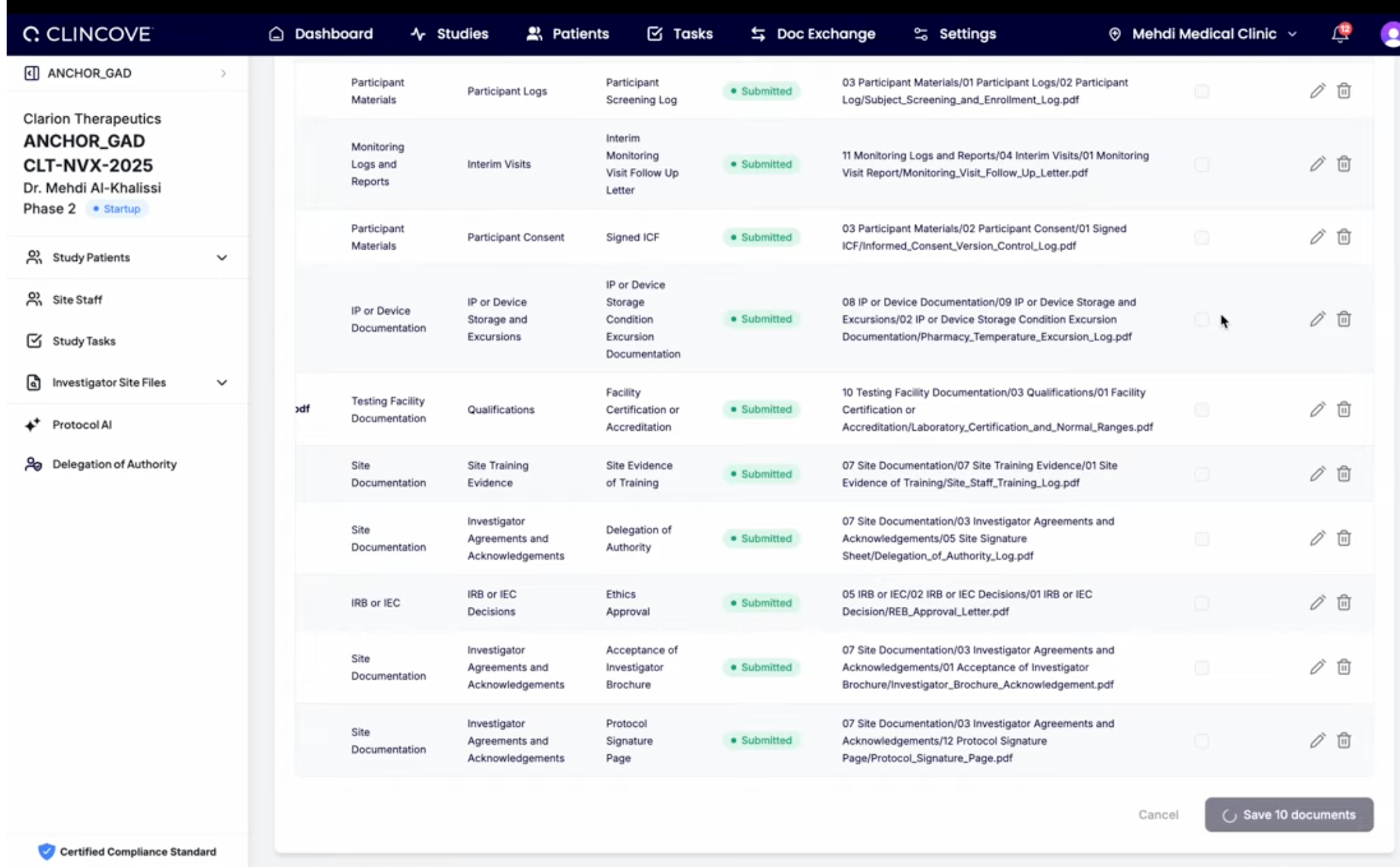
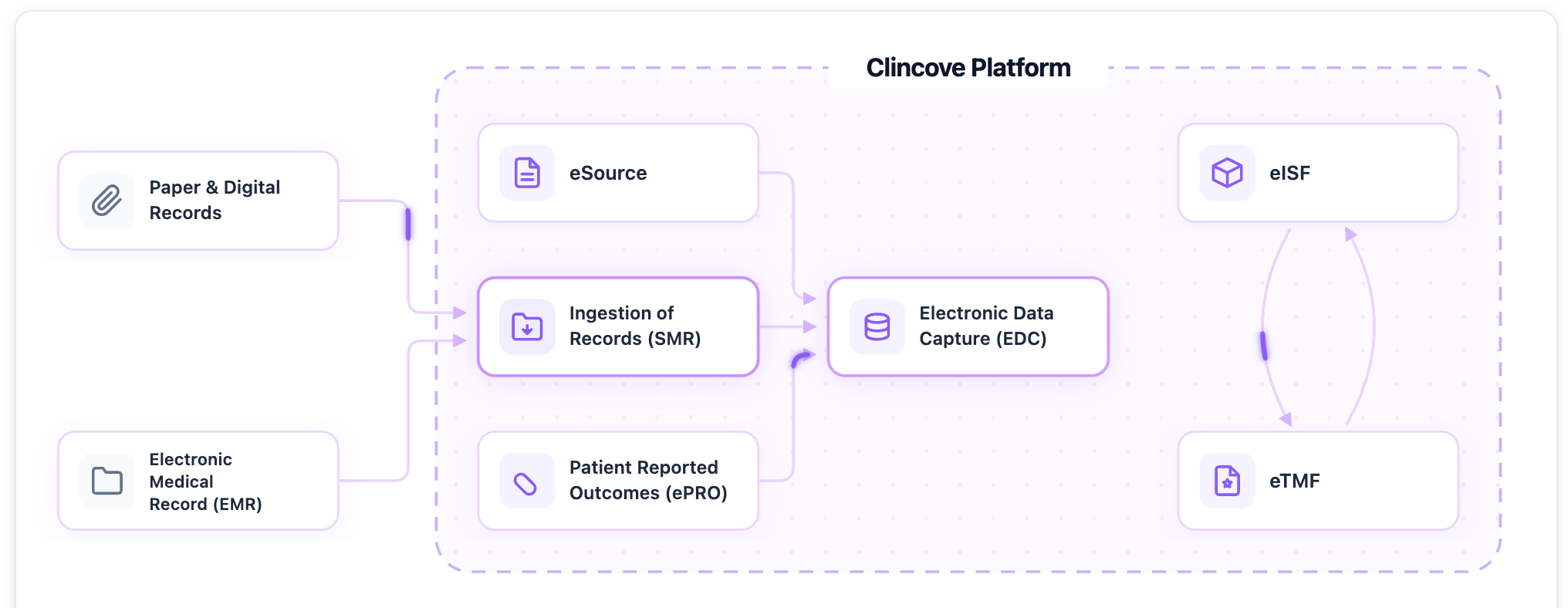
The same documents often need to exist in both the eTMF and eISF — signed protocols, IRB approvals, delegation logs, etc. When these are separate systems, documents get emailed back and forth, creating version control nightmares.
A unified document management platform that handles both eISF and eTMF eliminates this problem. Documents flow automatically between site and sponsor repositories, always in sync, always audit-ready. If you run clinical operations for sponsors, CRO or sites see how you can automate ISF and TMF Integration here.
Like what you see? Check out our 2-min Demo Video and see how Clincove unifies eISF and eTMF automatically in a single platform.
Continue Reading
Protocol AI: How We Built CRF (Case Report Forms) Auto-Generation - Modernizing EDC
A behind-the-scenes look at how Clincove’s Protocol AI reads protocol documents and automatically generates case report

Decentralized Trials in 2026: What’s Working and What Isn’t
DCTs promised to revolutionize clinical research. Three years post-pandemic, we look at what’s actually delivering resul

5 Red Flags in Your Clinical Trial Budget (And How to Fix Them)
Most trial budgets have hidden cost overruns baked in from day one. Learn to spot the five most common red flags before
Simplify clinical operations
Ready to modernize your clinical trials?
See how Clincove unifies EDC, eISF, eTMF, and eSource into one platform -- deploy in weeks, not months.