Site Activation in 2026: Contract to First Patient in 45 Days

Slow site activation is one of the most common reasons trials miss enrollment targets. Industry data shows average activation timelines of 3–6 months from contract execution to first patient in — but top-performing sites are doing it in 45 days.
Where the time goes
A typical site activation timeline breaks down like this:
- Contract and budget negotiation: 2–8 weeks
- IRB submission and approval: 2–4 weeks
- Regulatory document collection: 2–4 weeks
- System access and training: 1–2 weeks
- Site Initiation Visit: 1 week (scheduling often adds 2–3 weeks)
The biggest delays come from sequential processes that could be parallel, and from document collection bottlenecks.
How to cut activation to 45 days
- Parallelize everything. Start IRB submission, system setup, and document collection simultaneously — not sequentially.
- Use an eISF from day one. Electronic document management eliminates version confusion and enables remote collection. See the business case for eISF.
- Pre-qualify sites. Maintain a registry of site capabilities, PI CVs, and lab certifications that can be reused across studies.
- Standardize contracts. Use master service agreements with study-specific work orders instead of negotiating from scratch each time.
- Conduct virtual SIVs. When appropriate, virtual site initiation visits can eliminate weeks of scheduling delays.
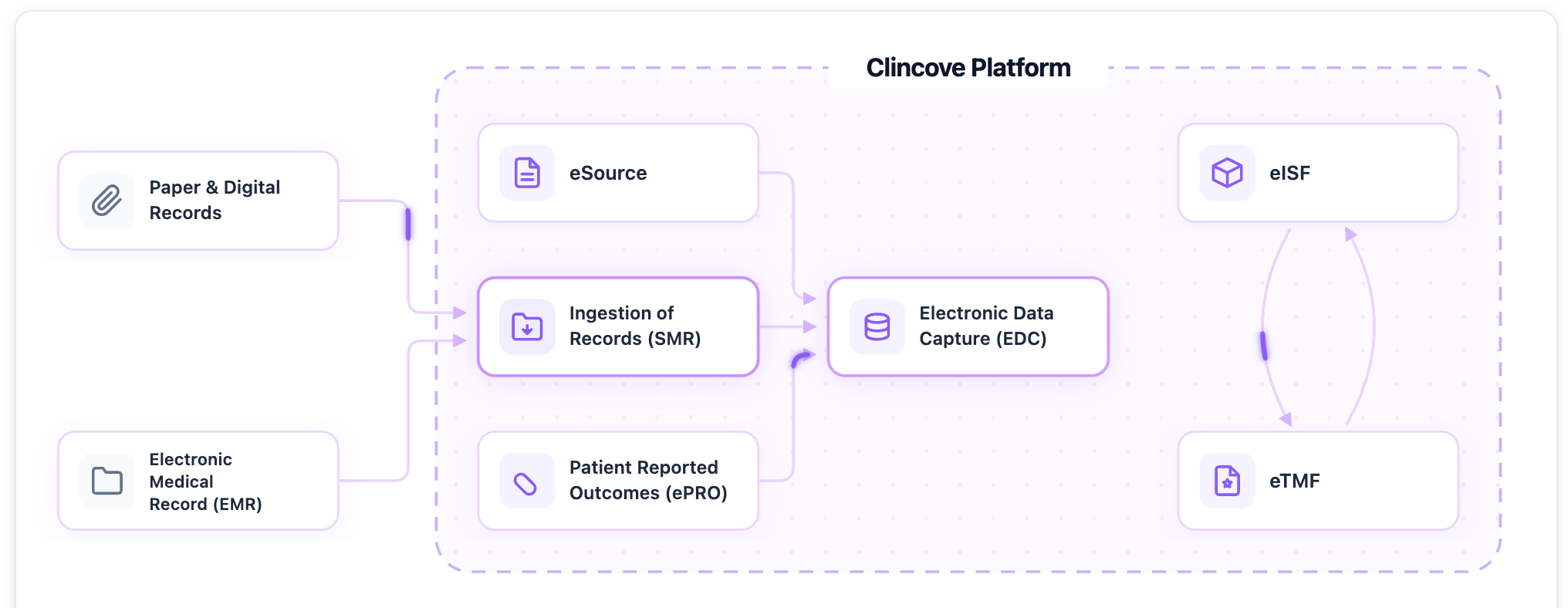
- Choose a unified platform. When EDC, eISF, and training all live in one system, there’s one access request instead of five.
The impact on enrollment
Every week saved in site activation is a week gained for Enrollment. For a 20-site trial, cutting activation from 12 weeks to 6 weeks effectively adds 6 weeks of enrollment capacity across the study.
For clinics launching their first trial, read our guide on going from zero to first patient in 60 days. For sponsors looking to accelerate across all sites, see how Clincove streamlines site activation.
Continue Reading

Decentralized Trials in 2026: What’s Working and What Isn’t
DCTs promised to revolutionize clinical research. Three years post-pandemic, we look at what’s actually delivering resul
Protocol AI: How We Built CRF (Case Report Forms) Auto-Generation - Modernizing EDC
A behind-the-scenes look at how Clincove’s Protocol AI reads protocol documents and automatically generates case report

5 Red Flags in Your Clinical Trial Budget (And How to Fix Them)
Most trial budgets have hidden cost overruns baked in from day one. Learn to spot the five most common red flags before
Simplify clinical operations
Ready to modernize your clinical trials?
See how Clincove unifies EDC, eISF, eTMF, and eSource into one platform -- deploy in weeks, not months.