How to Pass an FDA Inspection: Site Readiness Checklist

Table of Contents
An FDA inspection doesn’t have to be a source of anxiety. Whether it’s a routine surveillance inspection, a pre-approval inspection (PAI), or a for-cause investigation, the key to success is ongoing readiness — not last-minute preparation.
This checklist covers everything sites need to have in order when the FDA calls.
Before the inspector arrives
Documentation readiness
- Regulatory binder / eISF is current. All essential documents should be filed, signed, and dated. IRB approvals, protocol amendments, delegation logs, training records, and financial disclosures should all be up to date.
- Informed consent forms are complete. Every enrolled subject must have a signed ICF that matches the IRB-approved version at the time of their enrollment. Re-consent documentation for amendments must be present.
- Audit trails are accessible. If you’re using electronic systems, be prepared to demonstrate audit trail functionality and produce audit trail reports for specific subjects on request.
- Delegation log is current. Every person performing trial duties must be listed with their specific delegated responsibilities, qualifications, and training dates.
- Training records are documented. GCP training, protocol-specific training, and system training must be documented for every team member.
Source data readiness
- Source documents are identifiable and organized. The inspector will want to trace data from the CRF back to source. Whether your source is paper, EHR, or eSource, the path must be clear.
- EDC data is clean. Open queries should be resolved. Any pending data clarifications should be addressed before the inspection window.
- Protocol deviations are documented. Every deviation must be recorded, reported to the IRB per their requirements, and include corrective/preventive actions.
- Adverse events are reported. All AEs and SAEs should be properly documented, graded, and reported within the required timelines.
During the inspection
When the FDA inspector arrives:
- Be honest and direct. Answer questions truthfully. If you don’t know the answer, say so and offer to find out — don’t guess.
- Provide what’s requested, nothing more. Don’t volunteer information or documents that weren’t asked for.
- Take notes. Document every document the inspector reviews, every question asked, and every observation made.
- Have your team available. The PI, coordinator, and any delegated staff should be accessible during the inspection.
- Provide a private workspace. The inspector needs a quiet space to review documents.
The most common FDA inspection findings
- Informed consent issues — Missing signatures, wrong consent version used, inadequate documentation of the consent discussion
- Protocol deviations — Unreported or inadequately documented deviations from the approved Protocol
- Inadequate records — Missing source data, incomplete CRFs, or inability to trace data to its source
- Investigational product issues — Improper storage, incomplete accountability logs, or dispensing errors
- Part 11 deficiencies — Shared passwords, missing audit trails, or inadequate system validation
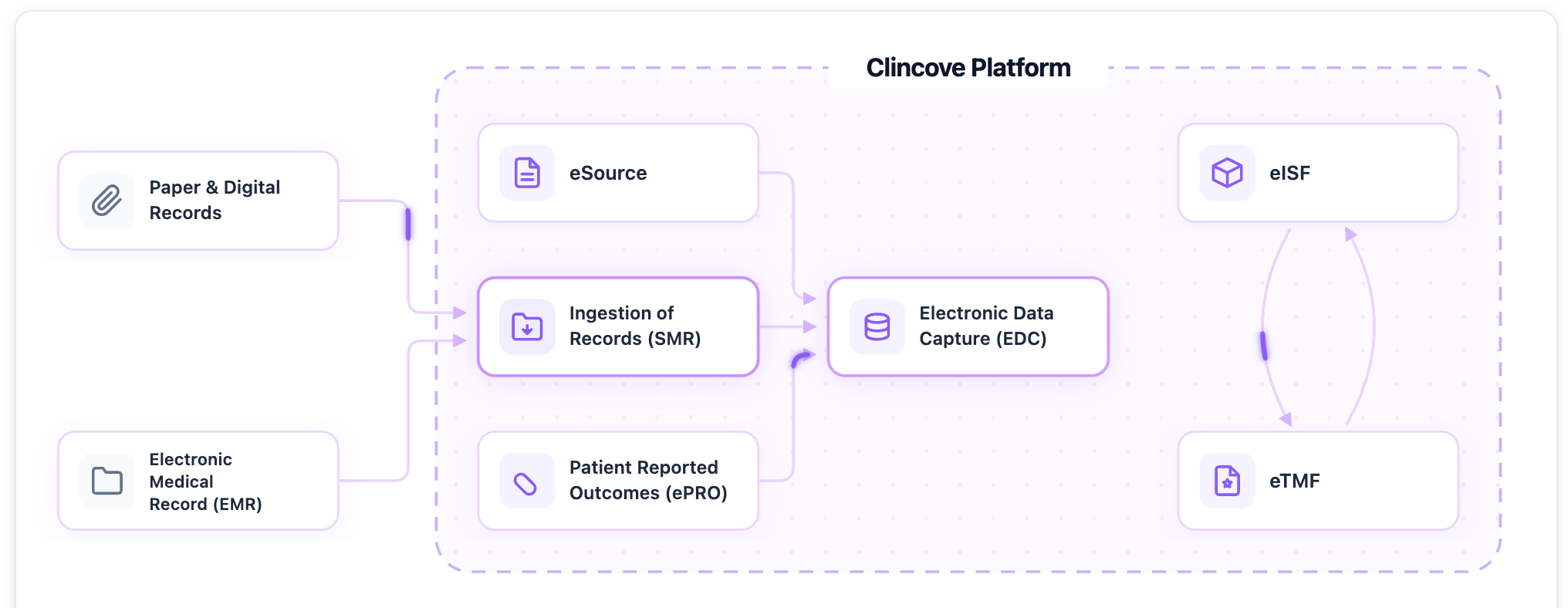
How technology makes inspections easier
Sites using a unified digital platform for their trial operations have a significant advantage during inspections:
- Electronic eISF provides instant access to all regulatory documents — no hunting through binders
- Built-in audit trails automatically capture the complete data history
- eSource eliminates source-to-CRF discrepancies entirely
- Real-time dashboards show compliance status at a glance
The best time to prepare for an inspection is before the trial starts — by choosing systems and processes that maintain compliance by design. See how Clincove keeps your site inspection-ready.
Continue Reading

Decentralized Trials in 2026: What’s Working and What Isn’t
DCTs promised to revolutionize clinical research. Three years post-pandemic, we look at what’s actually delivering resul
Protocol AI: How We Built CRF (Case Report Forms) Auto-Generation - Modernizing EDC
A behind-the-scenes look at how Clincove’s Protocol AI reads protocol documents and automatically generates case report

5 Red Flags in Your Clinical Trial Budget (And How to Fix Them)
Most trial budgets have hidden cost overruns baked in from day one. Learn to spot the five most common red flags before
Simplify clinical operations
Ready to modernize your clinical trials?
See how Clincove unifies EDC, eISF, eTMF, and eSource into one platform -- deploy in weeks, not months.