Protocol AI: How We Built CRF (Case Report Forms) Auto-Generation - Modernizing EDC
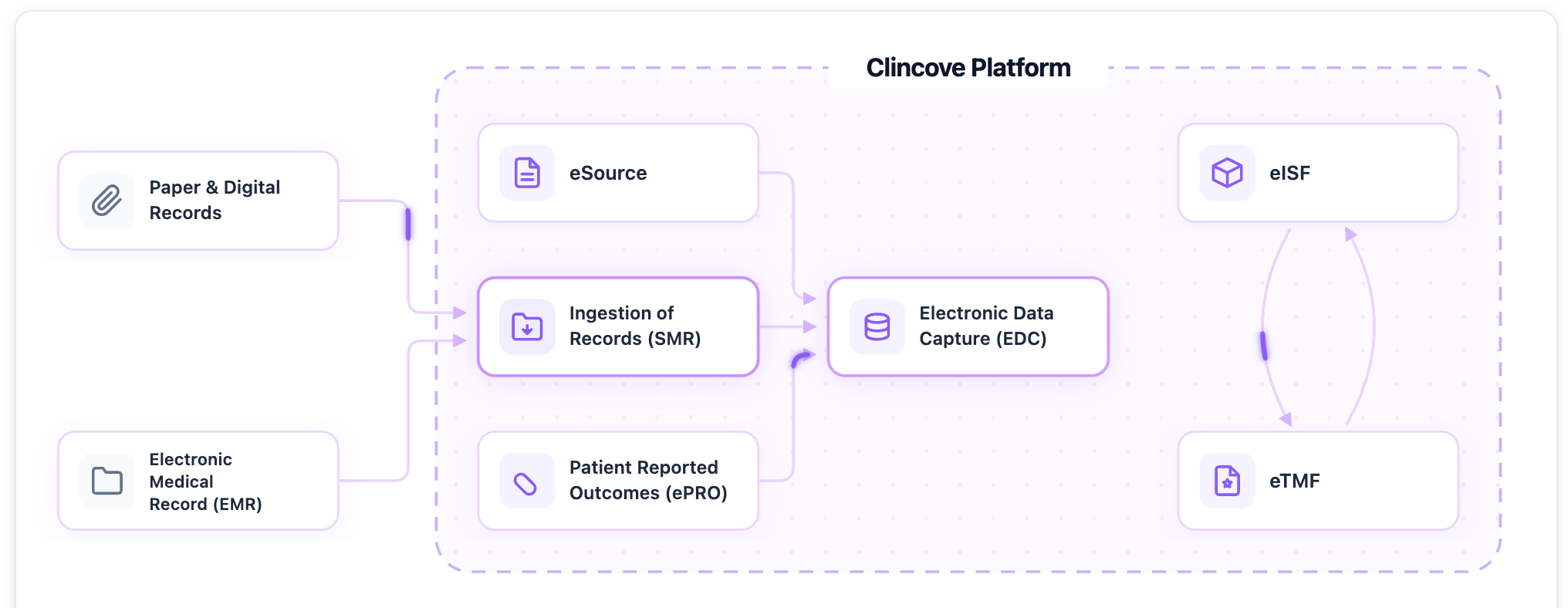
Table of Contents
Every clinical trial starts with a Protocol — a detailed document specifying the study design, endpoints, visit schedule, and data collection requirements. Translating that protocol into case report forms (CRFs) has traditionally been a manual, weeks-long process. We built Protocol AI to change that.
The traditional CRF design process
In a typical study, CRF design works like this:
- The medical team writes the protocol (4–12 weeks)
- A data management team reads the protocol and designs CRF pages (2–4 weeks)
- The sponsor reviews CRF drafts and requests changes (1–2 weeks, often multiple cycles)
- CRFs are built in the EDC system (1–2 weeks)
- User acceptance testing is performed (1 week)
Total: 8–20+ weeks from protocol to production CRFs. And that’s if everything goes smoothly.
What Protocol AI does
Protocol AI reads your protocol document and automatically generates a complete CRF package. Here’s how it works:
- Protocol parsing. The AI reads the protocol PDF and extracts the visit schedule, procedures, endpoints, inclusion/exclusion criteria, and data collection requirements.
- CRF generation. Based on the extracted requirements, it generates CDISC-aligned CRF forms with appropriate field types, edit checks, and visit mapping.
- Standards compliance. Forms are generated following CDASH standards and your organization’s library of approved CRF modules.
- Human review. The generated CRFs are presented for medical and data management review — they can be modified, approved, or regenerated with different parameters.
What we learned building it
Building Protocol AI taught us several lessons about applying AI to clinical trial operations:
- Protocols are surprisingly inconsistent. The same concept might be described five different ways across five protocols. The AI needs to understand intent, not just match keywords.
- Standards matter enormously. Without a strong CDISC/CDASH foundation, AI-generated CRFs would be technically correct but operationally useless.
- The 80/20 rule applies. AI can generate 80% of CRF content accurately from the protocol. The remaining 20% — complex conditional logic, sponsor-specific conventions, and edge cases — still needs human expertise.
- Speed creates value even when perfection isn’t possible. Getting a 90% complete draft in hours instead of weeks compresses the entire study startup timeline.
Results so far
Across studies using Protocol AI:
- CRF design time reduced from weeks to hours
- 30% fewer review cycles due to better first-draft quality
- Improved CDISC compliance in CRF design
- Faster study startup — first patient in sooner
See Protocol AI in action (2-Min Video Demo) — upload a protocol and watch it generate CRFs in real time.
Continue Reading
eTMF vs. eISF: What’s the Difference, Why It Matters & How to Automate Transfer & Filing
eTMF and eISF sound similar but serve very different purposes. Understanding the distinction is critical for choosing th

Decentralized Trials in 2026: What’s Working and What Isn’t
DCTs promised to revolutionize clinical research. Three years post-pandemic, we look at what’s actually delivering resul

5 Red Flags in Your Clinical Trial Budget (And How to Fix Them)
Most trial budgets have hidden cost overruns baked in from day one. Learn to spot the five most common red flags before
Simplify clinical operations
Ready to modernize your clinical trials?
See how Clincove unifies EDC, eISF, eTMF, and eSource into one platform -- deploy in weeks, not months.