How to Reduce Query Rates by 60% with eSource
Table of Contents
Clinical data queries are one of the most expensive and time-consuming aspects of data management. Industry benchmarks show average query rates of 15–25 queries per 100 CRF pages. Each query takes 5–15 minutes to resolve and costs $20–50 in site and CRO staff time. At scale, this adds up to millions of dollars per trial.
Where queries come from
The #1 source of queries is transcription error — data that was manually copied from source documents into the EDC and something went wrong:
- Typos and transposition errors (recording 120/80 as 120/08)
- Unit mismatches (recording weight in kg when the CRF expects lbs)
- Missing data (fields left blank during manual transcription)
- Inconsistent dates (recording a procedure date that doesn’t match the source)
- Illegible source data (handwritten notes that the coordinator can’t decipher during transcription)
How eSource eliminates the root cause
When you capture data electronically at the point of care using eSource, the transcription step — and all its associated errors — disappears entirely. The electronic capture IS the source document.
Real-world results from sites using Clincove’s eSource:
- 62% reduction in total queries compared to traditional source → EDC workflows
- 45% faster query resolution for the remaining queries (easier to investigate when audit trails are complete)
- 30% faster database lock due to cleaner data throughout the study
- 50% reduction in SDV time since source and CRF data are the same data
The math
For a typical Phase III trial with 500 subjects and 200 CRF pages per subject:
- Traditional approach: ~20,000–50,000 queries × $30 average resolution cost = $600K–$1.5M in query costs
- With eSource: ~8,000–20,000 queries × $20 average resolution cost = $160K–$400K in query costs
- Net savings: $440K–$1.1M per study
Getting started
eSource adoption doesn’t have to be all-or-nothing. Start with the highest-volume data domains (vitals, concomitant medications, consent documentation) and expand from there. Read our complete eSource guide for implementation details.
See Clincove’s eSource in action — capture clean data from the start.
Continue Reading

Decentralized Trials in 2026: What’s Working and What Isn’t
DCTs promised to revolutionize clinical research. Three years post-pandemic, we look at what’s actually delivering resul
Protocol AI: How We Built CRF (Case Report Forms) Auto-Generation - Modernizing EDC
A behind-the-scenes look at how Clincove’s Protocol AI reads protocol documents and automatically generates case report

5 Red Flags in Your Clinical Trial Budget (And How to Fix Them)
Most trial budgets have hidden cost overruns baked in from day one. Learn to spot the five most common red flags before
Simplify clinical operations
Ready to modernize your clinical trials?
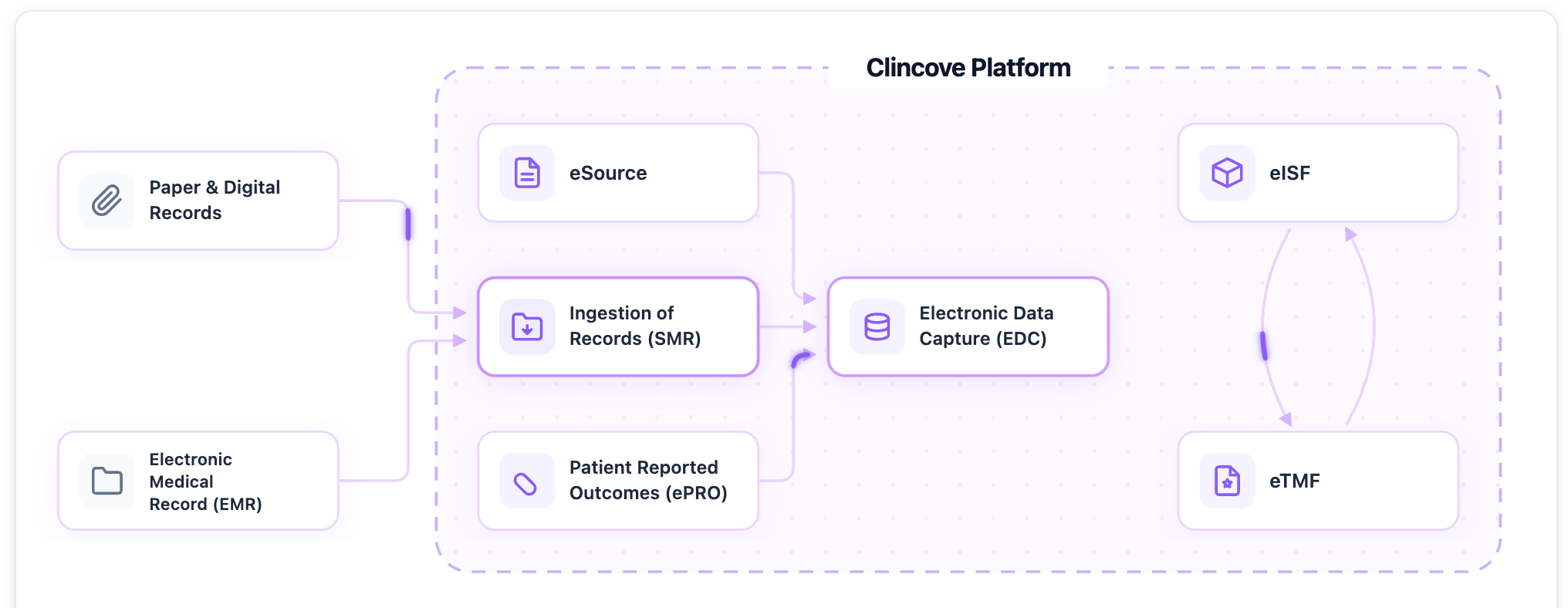
See how Clincove unifies EDC, eISF, eTMF, and eSource into one platform -- deploy in weeks, not months.