The CRA’s Guide to Remote Monitoring with Clincove

Table of Contents
Remote Monitoring has gone from pandemic workaround to permanent practice. For CRAs, this means new skills, new tools, and a fundamentally different approach to ensuring data quality and patient safety.
What remote monitoring actually looks like
Remote monitoring involves reviewing trial data, documents, and processes from a central location instead of traveling to the site. Under ICH E6(R2) and the FDA’s risk-based monitoring guidance, this is not just acceptable — it’s encouraged.
Key activities that can be done remotely:
- Source data verification. When sites use eSource, source and CRF data are the same — SDV can be performed entirely remotely.
- Document review. Consent forms, delegation logs, and regulatory documents in an eISF can be reviewed without traveling to the site.
- Query management. Identifying and resolving data discrepancies through the EDC system.
- KRI review. Monitoring site-level risk indicators via centralized dashboards.
- Site communication. Regular check-ins via video call to discuss open items and site needs.
What still requires on-site visits
- Investigational product accountability and storage verification
- Source document verification when source is paper or non-integrated EHR
- Process observation (how consent is actually conducted, not just documented)
- For-cause visits triggered by risk signals
- Relationship building with new sites or new coordinators
Best practices for remote CRAs
- Establish a regular cadence. Weekly or biweekly video check-ins with site coordinators keep communication flowing and issues from festering.
- Use structured review templates. Document what you reviewed, what you found, and what actions are needed — just as you would during an on-site visit.
- Prioritize by risk. Focus your remote review time on critical data points and sites flagged by KRI dashboards.
- Maintain the human connection. Remote monitoring can feel impersonal. Make time for non-agenda conversation. Ask how the coordinator is doing, not just how the data looks.
- Document thoroughly. Remote monitoring activities need the same level of documentation as on-site visits. If it’s not documented, it didn’t happen.
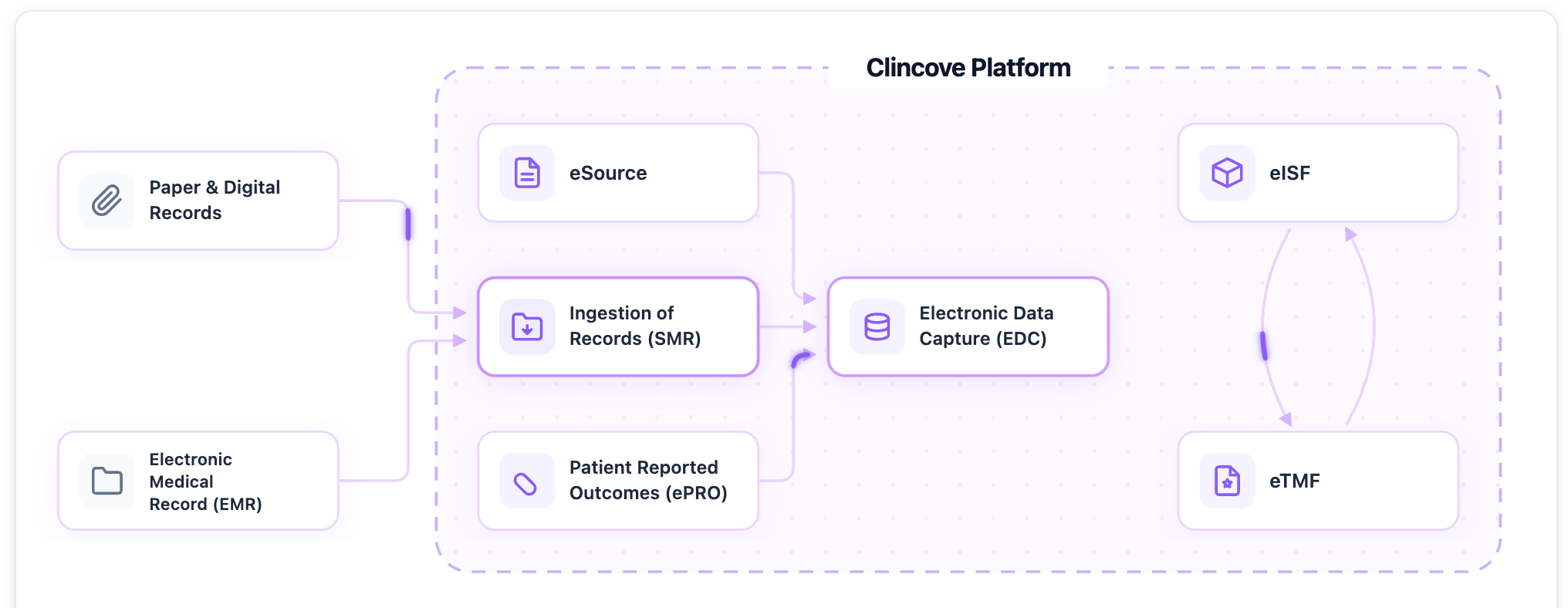
The technology foundation
Effective remote monitoring requires a platform that provides real-time access to study data, documents, and risk indicators from a single interface. When EDC, eISF, and eSource live in one unified system, CRAs can monitor effectively without switching between applications.
See how Clincove enables seamless remote monitoring for CRAs and clinical operations teams.
Continue Reading

Decentralized Trials in 2026: What’s Working and What Isn’t
DCTs promised to revolutionize clinical research. Three years post-pandemic, we look at what’s actually delivering resul
Protocol AI: How We Built CRF (Case Report Forms) Auto-Generation - Modernizing EDC
A behind-the-scenes look at how Clincove’s Protocol AI reads protocol documents and automatically generates case report

5 Red Flags in Your Clinical Trial Budget (And How to Fix Them)
Most trial budgets have hidden cost overruns baked in from day one. Learn to spot the five most common red flags before
Simplify clinical operations
Ready to modernize your clinical trials?
See how Clincove unifies EDC, eISF, eTMF, and eSource into one platform -- deploy in weeks, not months.